By Wilhelm Pfleging, Melanie Mangang, Yijing Zheng, Peter Smyrek and Johannes Pröll
Introduction
Thick film anodes and cathodes with thicknesses ranging from 20-300 µm, in state-of-the-art and future lithium-ion cells are complex multi-material systems consisting of defined material components, grain sizes, porosities and pore size distributions in the micrometer and submicrometer ranges. State-of-the-art cells with pouch cell geometry for high power applications consist of thick film electrode stacks with capacities up to 40-50 Ah.
The development of three-dimensional (3D) cell architectures for electrodes in lithium-ion batteries is a promising approach to overcome problems like 1-dimensional lithium-ion diffusion, inhomogeneous current densities, power losses, high interelectrode ohmic resistances as well as mechanical stresses due to high volume changes resulting from lithium-ion insertion and deinsertion. By applying 3D battery architectures, one can achieve large areal energy capacities while maintaining high power densities at the same time. This feature is important, e.g., for thin film batteries where the lithium-ion diffusion is limited by the thickness of the compact film. A common approach for realization of 3D architectures in electrodes is the structuring of the substrate or current collector. An increased active surface achieved by 3D electrode architectures can induce large areal energy densities. Unfortunately, this approach is in a very early stage of development and in general it is not feasible for state-of-the-art electrodes.

At the Karlsruhe Institute of Technology (KIT), a new process for the generation of 3D electrode designs has been created by developing two processes; 1.) Laser-assisted self-organized structuring (Fig. 1) and 2.) direct structuring of tape cast electrodes [1-3].
In each case, the laser structured electrodes exhibit a significant improvement in liquid electrolyte wetting as well as in electrochemical performance after laser treatment. During the manufacturing process of lithium-ion cells, liquid electrolyte filling is a cost- and time-consuming process. Insufficient electrolyte wetting in turn can lead to unexpected cell failure under challenging cycling conditions. At KIT, a cost efficient laser-based technology for the realization of 3D architectures in thick-film tape-cast electrodes was developed to accelerate the wetting process and to also shorten the time-span for cell manufacturing (Fig. 2).
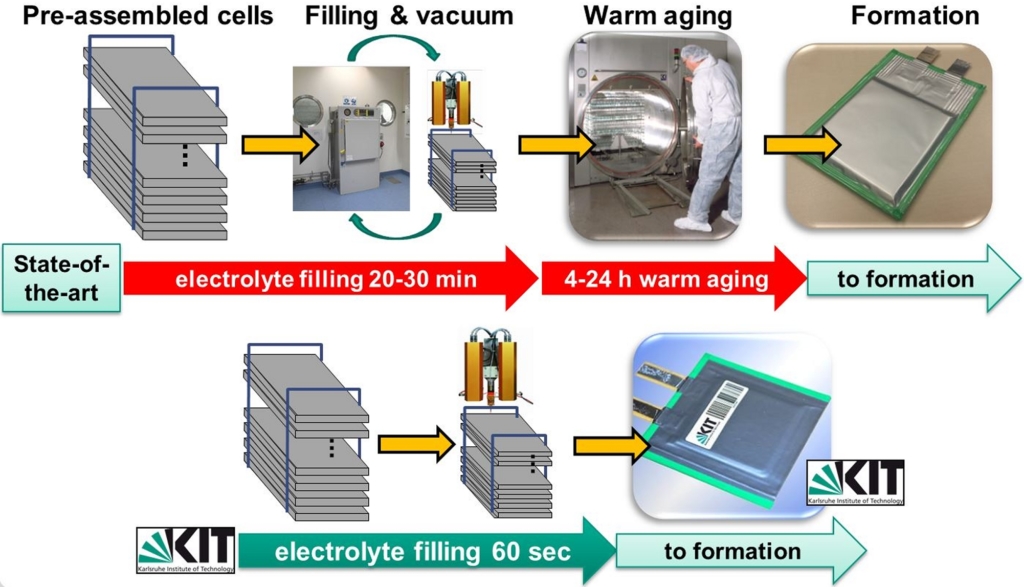
In addition, an improved cell operation with extended life-time and increased capacity retention at high charging and discharging currents could be achieved. For the development of advanced laser processes in battery manufacturing, a complete lithium-ion cell manufacturing process cycle has been built-up which includes electrochemical characterization of lithium-ion cells (Fig. 3).

Experimental Setup
Different types of electrode materials were already investigated such as LiCoO2 (LCO), LiMn2O4 (LMO), SnO2 (SnO), fluorine doped SnO2 (FTO), Li(NiMnCo)O2 (NMC), silicon (Si), graphite (C) and LiFePO4 (LFP). Thin films as well as thick films were applied. Thick film electrodes are composite materials which consist of active material, carbon black, graphite and binder. All lithium-ion cells were assembled either in an argon-filled glove box or in a dry room. An ultrafast fiber laser system (Tangerine, Amplitude Systèmes, France), a ns fiber laser system (YLPM, IPG Photonics, Germany), or an excimer laser system (ATLEX-1000-I, ATL Lasertechnik GmbH, Germany) were used to manufacture 3D architectures into the thin or thick film electrode layers.

The Results
In general, electrolyte filling of lithium-ion cells is realized by time and cost consuming vacuum and storage processes at elevated temperatures. Nevertheless, by applying state-of-the-art electrolyte filling processes, insufficient wetting of electrode and separators is one drawback resulting in a certain production failure rate accompanied with a lowered cell capacity or a reduced cell life-time. Laser structuring has been developed for the formation of capillary micro-structures in thick film tape-cast electrodes which resulted in the acceleration of electrolyte wetting in comparison to unstructured electrodes (Fig. 4). The removal of the complete electrode material from the ablation zone delivers the most efficient capillary transport [4].
For the formation of capillary structures, ns-laser ablation as well as ultrafast laser processing was investigated.
For ns-laser radiation (l=1064 nm, pulse length 200 ns) the laser beam energy is absorbed at the material surface and, due to heat conduction, the temperature of the surrounding composite material increases. The binder material for tape-cast electrodes (~5 wt%) is PVDF which has a low decomposition temperature in the range of 250–350° C [5]. Therefore, the PVDF binder matrix spontaneously evaporates and active particles are removed from the laser beam interaction zone.

With ns-laser radiation, structure widths of about 40-55 µm can be achieved (Fig. 5a & 5b). The current collector for cathodes are made of aluminium with a thickness of 20 µm and for anodes they consist of copper with a thickness of 10 µm. Laser structuring with a ns-laser can be realized without damage of the current collector (Fig. 5a). Laser structuring can be realized even for double-side coated aluminium substrates, which is a required processing step for process up-scale for manufacturing of lithium-ion cells with high capacities [4].
Nanosecond laser ablation is not appropriate for each type of electrode material. For example, ns laser structuring of LFP electrodes always leads to melt formation and therefore to an undesired modification of the active material. Furthermore, the ablation efficiency of LFP increases by a factor of 3 by using femto- or pico-second laser ablation in comparison to ns-laser ablation [6]. Another aspect is the loss of active material due to the ablation process. For the application of structured foils in batteries, it is important to reduce the amount of ablated material which in turn means that small capillary widths and high aspect ratios are preferred. By using ultrafast laser ablation it could be shown that the aspect ratio could be significantly increased (Fig. 5c & 5d) and that the loss of active material can be reduced from 20 percent down to values below 5 percent [7].
Capacity retention and cell life-time can be illustrated by plotting the cell voltage as function of discharge capacity for different cycle numbers. For the lithium-ion cell with the structured NMC electrode, the 80 percent capacity limit of the initial discharge capacity is reached after 2290 cycles (Fig. 6). While the cell life-time for the lithium-ion cell with unstructured electrodes is reached after 141 cycles. Furthermore, the discharge capacity of the cell with the laser-structured NMC electrode reaches a value of 108 mAh/g after 2290 cycles indicating that efficient liquid electrolyte transport due to micro capillary structures improves the electrochemical performance for cell without cost- and time-consuming storage procedures.
![Figure 6. Cell voltage versus discharge capacity for pouch cells with laser-structured (right) and unstructured (left) NMC electrodes and without storage [4]](https://www.laserstoday.com/wp-content/uploads/2016/04/Fig-6-1024x400.jpg)
Summary & Conclusion
A new technical approach of using laser-generated capillary structures in electrode materials was presented. This technology can be applied in order to increase cell reliability during the production process, to shorten production times of lithium-ion cells as well as to increase the cell life-time during cycling. Due to an improved cycle life-time and increased capacity retention, the use of high power batteries in 2nd life applications becomes interesting. Cost-efficient ns fiber lasers can be applied for carrying out the structuring process for several types of electrode materials. Nevertheless, regarding the structuring of LFP, a further reduction of active mass loss, and an up-scaling of the structuring process, the use of ultrafast laser processing becomes necessary.
References
- J. Pröll, H. Kim, A. Piqué, H.J. Seifert, W. Pfleging, J. Power Sources, 255(0) (2014), 116-124.
- J.H. Park, R. Kohler, W. Pfleging, W. Choi, H.J. Seifert, J.K. Lee, RSC Adv., 4(9) (2014), 4247-4252.
- R. Kohler, J. Pröll, M. Bruns, S. Ulrich, H.J. Seifert, W. Pfleging, Appl. Phys. A, 112(1) (2013), 77-85.
- W. Pfleging, J. Pröll, J. Mater Chem A, 2(36) (2014), 14918-14926.
- J. Choi, E. Morikawa, S. Ducharme, P.A. Dowben, Mater Lett, 59(28) (2005), 3599-3603.
- M. Mangang, H.J. Seifert, W. Pfleging, J. Power Sources, 304 (2016), 24-32.
- P. Smyrek, J. Pröll, H.J. Seifert, W. Pfleging, J. Electrochem Soc, 163(2) (2016), A19-A26.
Wilhelm Pfleging, Yijing Zheng, Peter Smyrek and Johannes Pröll are all with the Karlsruhe Nano Micro Facility in , Germany. They are joined by Melanie Mangang in their work at Karlsruhe Institute of Technology (KIT).





